Functional Analysis of the MAG2 Body
Seed storage proteins are synthesized as precursors on the endoplasmic reticulum (ER) and then transported to protein storage vacuoles, where they are processed into mature forms. MAG2 body is a novel structure found in cells, containing storage protein precursors and endoplasmic reticulum molecular chaperones. It has a core part with high electron density and a matrix part with low electron density. The core fraction consists of precursors of 2S albumin, and the matrix fraction contains precursors of 12S globulin as well as immunoglobulin heavy chain-binding protein and protein disulfide isomerase.
Lifeasible provides functional analysis of the MAG2 body, including storage protein precursors and molecular chaperones, to help our customers worldwide in the field of plant science. Our platform is equipped with cutting-edge facilities and professional experts to support research. Here, we provide various services according to customers' demands.
MAG2 Body Stores Protein Precursors
- MAG2 Body can accumulate precursors of two major storage proteins, 2S albumin and 12S globulin, in seeds.
- Lifeasible helps our customers study this process through the following methods. To understand the molecular mechanism of MAG2 Body accumulation of protein precursors, we create plant mutants of the MAG2 gene. Immunoblotting is used to verify the specific reaction of the antibody with the main storage protein, 12S globulin, and 2S albumin.
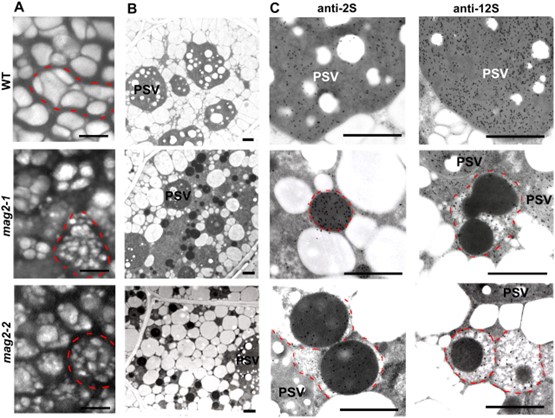 Fig.1. MAG2 Arabidopsis mutant seeds develop a number of novel structures that accumulate storage proteins. (Li L., et al., 2006)
Fig.1. MAG2 Arabidopsis mutant seeds develop a number of novel structures that accumulate storage proteins. (Li L., et al., 2006)
- In terms of morphology, laser scanning confocal microscopy is used to observe the cellular structure of the mutant, especially the protein storage vacuoles (PSV) morphology. Finally, to determine the subcellular localization of the precursor protein accumulated in mutant seeds, we generate specific antibodies against an N-terminal propeptide of 2S3, which is removed by VPE proteolysis.
MAG2 Body Stores Molecular Chaperones
- MAG2 body also can accumulate molecular chaperones in plant endoplasmic reticulum, including immunoglobulin heavy chain-binding protein and protein disulfide isomerase.
- We provide experimental protocols to study this function of the MAG2 body. To identify protein components abnormally accumulated in seeds, proteins in wild-type and mutant seeds are separated on 2D gels for proteomic analysis. Further, Coomassie blue staining is performed to observe the appearance of visually different signal intensities on the gel.
- In addition, with immunogold analysis of mutant seeds, we obtain the location where the MAG2 body accumulates ER chaperones.
Lifeasible provides cost-effect, high quality, and hassle-free services to our clients worldwide. We provide our clients with direct access to our experts and prompt response to their inquiries. If you are interested in our services or have any questions, please feel free to contact us or make an online inquiry.
Reference
- Li L, et al. (2006). "MAIGO2 is involved in exit of seed storage proteins from the endoplasmic reticulum in Arabidopsis thaliana." Plant Cell. 18 (12), 3535-47.
For research or industrial raw materials, not for personal medical use!
 Fig.1. MAG2 Arabidopsis mutant seeds develop a number of novel structures that accumulate storage proteins. (Li L., et al., 2006)
Fig.1. MAG2 Arabidopsis mutant seeds develop a number of novel structures that accumulate storage proteins. (Li L., et al., 2006)